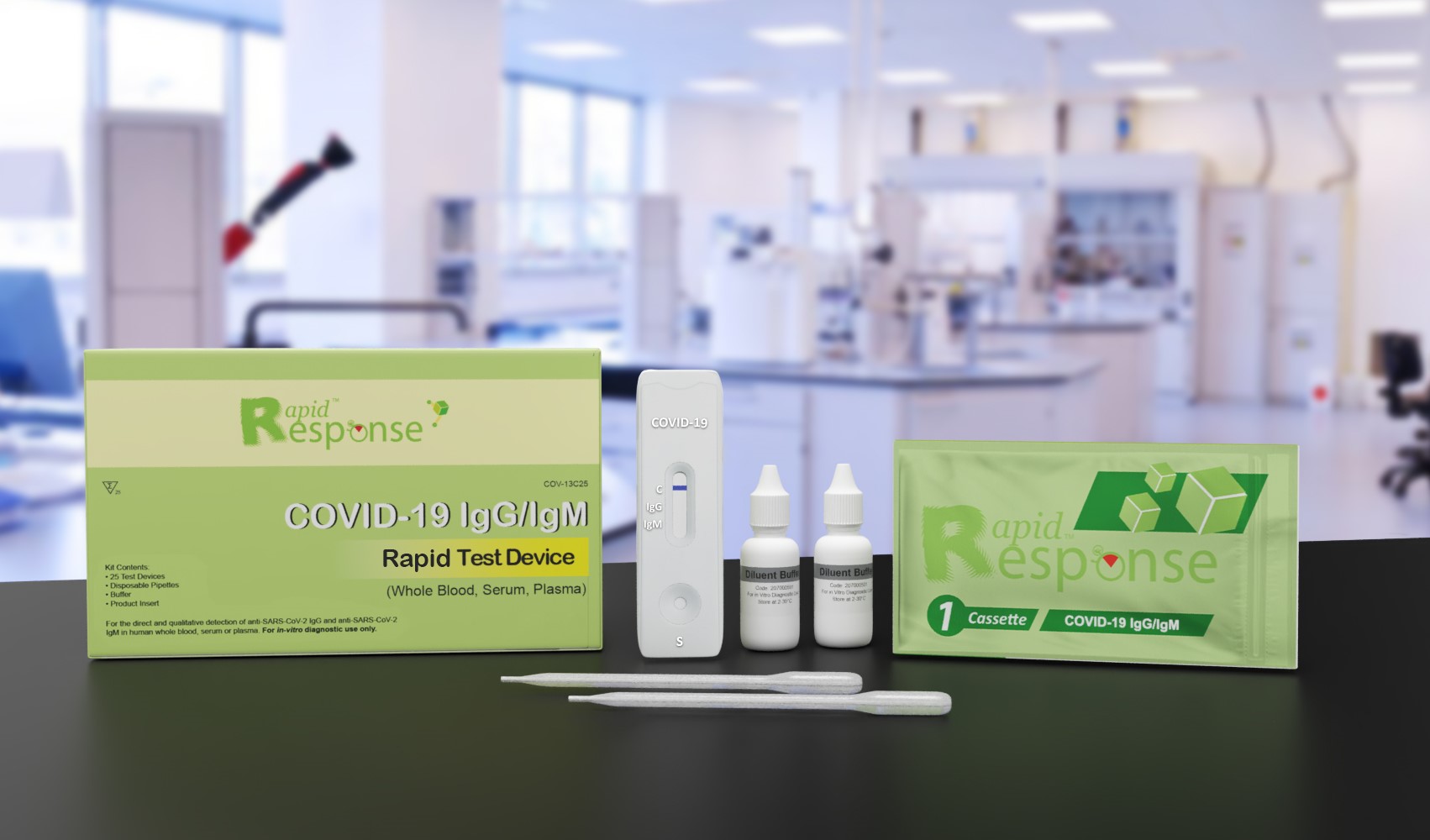
COVID-19 IgG/IgM Test
Product Information
Product Code: COV-13C25Sample: Whole Blood / Serum / Plasma
Format: Cassette
Quantity: 25 Tests/Kit
Sensitivity: IgM: 93.7%, IgG: 98.8%
Specificity: COVID-19 virus IgG and IgM antibody
Time to result: 15 minutes
Storage Condition: 2-30°C/35.6-86°F
Test Principle: Immunochromatographic Lateral Flow Assay
Contents
Test CassettesDisposable Pipettes
Buffer
Product Insert
Product Description
The Rapid Response™ COVID-19 IgG/IgM Rapid Test Device is an in vitro immunoassay for the direct and qualitative detection of anti-SARS-CoV-2 IgM and anti-SARS-CoV-2 IgG in human whole blood (including venous whole blood and capillary whole blood), serum, or plasma. At the Point of Care setting, this test is only authorized for use with fingerstick whole blood specimens. This test is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection. The Rapid Response™ COVID-19 IgG/IgM Rapid Test Device should not be used for screening patients or to diagnose or exclude acute SARS-CoV-2 infection. At this time, it is unknown for how long antibodies persist following infection and if the presence of antibodies confers protective immunity. A negative or non-reactive result for an individual subject indicates absence of detectable COVID-19 virus antibodies. However, a negative or non-reactive rest result does not preclude the possibility of exposure to or infection with COVID-19 virus. False positive results may occur due to cross-reactivity from pre-existing antibodies or other possible causes. Due to the risk of false positive results, confirmation of positive results should be confirmed using a second, different IgG/IgM detection method. Laboratories are required to report all results to the appropriate public health authorities. The test is for professional use only. This assay is not intended for home testing (or self-testing). The Rapid Response™ COVID-19 IgG/IgM Rapid Test Device may detect a response to vaccination.
Pursuant to section 5 of the Interim Order Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19, made by the Minister of Health on March 18, 2020, the Rapid Response™ COVID-19 IgG/IgM Rapid Test Device is now authorized for sale or importation in Canada.
Clinical Evaluation Data for Venous Whole Blood and Serum Samples:
IgG Detection: Relative Sensitivity 94.6% (CI: 84.2%-98.6%), Relative Specificity: 100% (CI: 96.5%-100%)
IgM Detection: Relative Sensitivity 92.9% (CI: 81.9%-97.6%), Relative Specificity: 98.1% (CI: 93.3%-99.5%)
Upload test results to your database with our RapidReader App. Learn More
Rapid Response® COVID-19 IgG / IgM Test Cassette
French version/version française
Safety Information
The test is for in vitro diagnostic use for professional use only.
For information on the IFU and purchase orders please contact your local sales representative or contact us at covid19@btnx.com or 1-888-339-9964.
For additional information regarding any of our products, please feel free to Contact Us